Introduction: In order to minimize chance of amputation due to actinomycetoma, it is important to correctly identify the causative agents. Microscopic examination of grains is not definite and further confirmatory diagnostic tests are needed. This study aims to investigate the prevalence of actinomycetoma and to explore the usefulness of strb1 gene in the diagnosis of the disease. Materials & Methods: The present study is a prospective cross-section laboratory-based study in which clinical samples (n = 100) from patients with mycetoma lesions were collected. The samples were cultured on Lowenstein Jensen and glucose yeast extract agar media. Grown colonies were initially identified using Gram’s stain, Ziehl Neelsen stain, and selected biochemical reactions. Confirmation was done by the analysis of polymerase chain reaction amplified strb1 gene. Results: Actinomycetoma was represented by a high ratio (12%) among the study population. Nine out of the 12 isolates (75%) were found to belong to the genus Streptomyces; whereas three isolates (25%) were identified as Nocardia spp. on the basis of phenotypic and mycolic acid contents. Conclusion: It could be concluded that actinomycetoma exists with significant prevalence (12%) among patients investigated in the present study. Streptomyces is the most important etiological agent of actinomycetoma compared to Nocardia. Keywords: Actinomycetoma, mycolic acids, Nocardia, strb1 gene, Streptomyces, Sudan
Mycetoma is a chronic specific granulomatous subcutaneous inflammatory disease that is produced by various causative agents that can be true fungi or aerobic actinomycetes of the genera Actinomadura, Nocardia, and Streptomyces. Actinomadura spp. commonly occurs in some countries and can produce as much as 50% of the total cases of mycetoma. [1] Actinomycetoma is one of the most neglected diseases and is considered a major health problem in several parts of Sudan. [2],[3],[4] Geographical distribution of mycetoma causative agents show considerable variations which could be explained by several environmental factors, specially the rainfall. [1],[5] The major agents of aerobic actinomycetes are: Nocardia brasiliensis, Actinomadura madurae, and Streptomyces somaliensis. [6],[7] Streptomyces sudanensis has recently being described as one of the etiological agents of actinomycetoma. [8] Many isolates belong to the genus Streptomyces were described to cause actinomycetoma in Sudan and were found to have variable antibiotic profiles which make in vivo treatment very difficult. [9] People at risk are mainly males and farmers in rural areas due to their nature of contact with microorganism, which lives as saprophyte in soil and other natural ecological niches. Mycetoma is not considered to be transmissible from person to person or from animal to person. There are some conflicting reports about the role of the immune status of susceptible population. Some investigators reported partial impairment of the cell-mediated immune response in patients severely infected or not responding to medical treatment. [10] The infection progresses slowly over a long period of time without painful manifestation and ultimately may extend to deep tissues and bones leading to deformity of the affected site and disability. [2],[11] It is important to identify the causative agent in order to develop a correct plan of treatment. Direct examination of grains may be useful in determining the type of mycetoma. Unlike fungal grains, crushed bacterial grain show fine filament, which can be stained according to Gram’s. Isolation of mycetoma agents can be difficult, especially in case of actinomycetoma. Culture required deep surgical biopsy containing grain. Biopsies without grains are not suitable for diagnosis. [12] It is important to identify the causative agents of actinomycetoma and to conduct regular hospital-associated surveillance of actinomycetoma infections. Also in vitro sensitivity testing of the causal agents is needed to prompt the treatment alone or in conjunction with surgery to reduce chance of amputation and prolong patient hospitalization due to actinomycetoma. This study aims to investigate the prevalence of actinomycetoma and to explore the usefulness of strb1 gene in the diagnosis of the disease.
Ethical clearance The ethical approval of this study was obtained from the Ethical Committee of Federal Ministry of Health and the Ethical Committee of Research Council of the Sudan University of Science and Technology. Written consent was obtained from every patient before being enrolled in the study. Study design The study was a cross-sectional study. Clinical specimens (n = 100) including grains from sinuses discharges and incision biopsies were collected. Mycetoma patients attending Mycetoma Research Center, Soba Teaching Hospital, Khartoum; Wad Madeni Teaching Hospital, and Um Rawaba Hospital from November 2009 to December 2010 were included in this study. Grains were taken from sinuses (lesions) and washed several times with sterile normal saline, and were then inoculated onto Lowenstein Jensen (LJ) slopes and glucose yeast extract agar (GYEA) media. Plates were incubated at 37°C and examined after 7, 14, and 21 days for the presence of microbial growth. Identification of isolates Typical and well-isolated actinomycetes-like colonies from the primary cultures were purified by sub-culturing on LJ, trypticase soya agar (TSA), and GYEA. Isolates were identifies on the basis of colony morphology and confirmed using selected phenotypic and mycolic acids analysis. Phenotypic characterization Isolates were tentatively identified as member of the genus Streptomyces on the basis of selected phenotypic criteria. [13] The cultural and microscopic features of the genus Streptomyces are: aerobic growth, Gram-positive, non-acid-alcohol-fast, non-motile actinomycete which forms extensively branched, light-yellow substrate mycelia on a variety of media with or without aerial hyphae, with or without diffusible pigments on the media. In the present study, phenotypic clusters of isolates were identified mainly on colony color and presence of diffusible pigments. Mycolic acid profiles The extraction of mycolic acids and thin layer chromatography (TLC) analysis of extracted mycolates was done as described by previously. [14] Amplification of strb1 gene DNA extraction from cultures was done by the standard phenol chloroform method according to Chomczynski and Sacchi. [15] Amplification of strb1 region (490 bp) [16] was performed with a set of primers having the following sequence: Forward primer: (5-TGAGCCTTGTAAGCGTCCAC-3) and reverse primer: (3-TTCATGCCGTGCTTCTCCAG-5). Master mix was prepared for 10 reactions according to Huddleston et al. [16] S. somaliensis DSM 41607 isolate was used as positive control. The negative control contained a reaction mixture without template DNA. PCR program was conducted with an initial 10 min denaturation step at 95°C for 1 cycle, followed by repeating cycles of denaturation (94°C for 30 s), annealing (30 s at 55°C), and extension (30 s at 72°C) for 40 cycle, followed by 5 min of final extension step at 72°C. The PCR products were electrophoresed on 1.5% agarose gel, containing ethidium bromide (10 μg/ml) to ensure that a fragment of the correct size had been amplified.
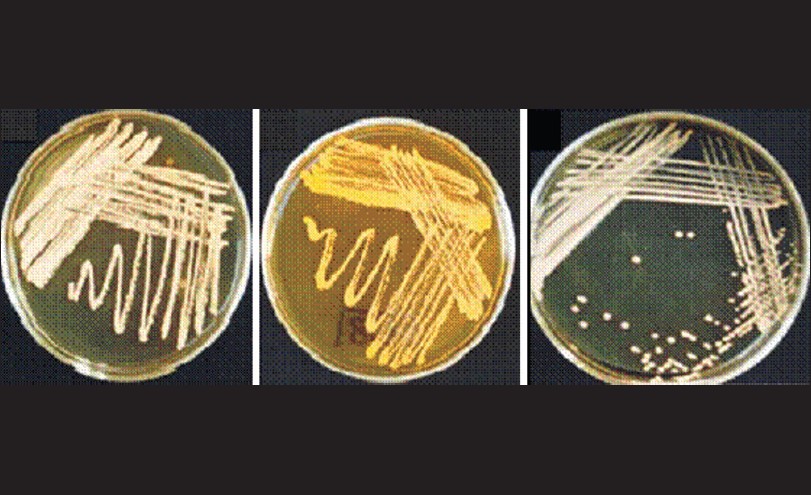
Demographic data In the present study, 100 patients who were presented with signs of mycetoma were enrolled. The majority of the patients were males (73%), compare with females (17%). Patients were categorized into three age groups: Group 1 (<20 years; 17%), group 2 (21-49 years; 57%), and group 3 (>50 years; 26%). The majority of the patients were farmers (64%) followed by pastoralists (23%), students (4%), and people with other minor jobs (13%). Phenotypic properties Eighty eight of the samples were tentatively identified as eumycetoma (88%) depending on the texture of the grains (course and black grains). The rest of the isolates (n = 12) appeared with different coloration and were tentatively classified as actinomycetoma. Colony morphology and different biochemical tests were used to identify these isolates. Nine out of the 12 actinomycete isolates (75%) showed typical appearance of Streptomyces spp. The results confirmed that three of the 12 isolates fit in the genus Nocardia given their phenotypic properties after 3-4 days aerobic growth on GYEA medium at 37°C. The distinguishing features are: formation of glabrous, rough, adherent, waxy, or dry chalky colonies. Colony colors range from cream-yellow, orange to gray-white with or without diffusible brown back pigments [Figure 1]. Microscopically they were characterized by the formation of branching filamentous gram positive bacteria which can fragments into short rods and coccobacillary forms.
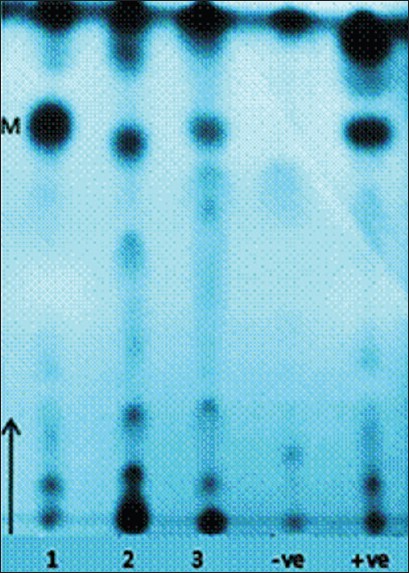
Thin layer chromatography analysis TLC analysis of mycolic acids extracted from the three suspected Nocardia spp. revealed characteristic nocardo-mycolates with Rf values equivalent to references Nocardia species [Figure 2].
PCR analysis Seven of the nine Streptomyces spp. showed 490 bp bands equivalent to strb1 region sequence [Figure 3]. The remaining two strains were identified on the basis of phenotypic characters.
The present study substantiates previous studies that actinomycetoma in Sudan can be caused by at least by two types of actinomycetes: Streptomyces spp. and Nocardia spp. The prevalence of these organisms was found to be 12%. The phenotypic and genotypic data show that 75% of these organisms were Streptomyces spp., whereas, 25% were Nocardia spp. Occurrence of Streptomyces among actinomycetoma patients in Sudan has long being recognized. [2],[3],[4],[8] But to our knowledge this is the first report to isolate Nocardia spp. from actinomycetoma patients in Sudan. Three strains isolated in the present study showed distinct phenotypic profiles in accordance with those of the genus Nocardia. Further taxonomic studies are needed to clarify the taxonomic position and fully identify these isolates. The results of the present study underline the importance of isolating and characterizing the causal agents of actinomycetoma in an appropriate clinical setting as it is evident that the color and morphology of grains isolated from biopsy material is not sufficient for the accurate identification of actinomycetes that cause mycetoma. In view of the present findings, we advocate the use of different phenotypic, chemotaxonomic, and genotypic properties to recognize the causative agents of mycetoma. Similar recommendations have been given in the past. [8],[9],[17]
In many developing countries no simple test is currently available for the diagnosis of mycetoma other than clinical assessment and the invasive procedure of surgical biopsy to take grain from non-open sinuses. When appropriate diagnostic tests are done the identity of the causative agents could be revealed. It could be concluded that actinomycetoma exists with significant prevalence (12%) among patients investigated in the present study. Streptomyces is the most important etiological agent of actinomycetoma compared with Nocardia. PCR could be a quick alternative given its high sensitivity and specificity, which may encourage its use rather than the tedious and time consuming conventional methods in diagnosis of mycetoma in the Sudan.
The authors acknowledge Prof. A.H. Fahal (Mycetoma Research Center, University of Khartoum) who facilitated samples collection and Prof. M. Goodfellow (School of Biology, University of Newcastle) for the supply of reference strains.
Source of Support: This work have been partially funded by Sudan University of Science and Technology.,, Conflict of Interest: None
[Figure 1], [Figure 2], [Figure 3] |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||