| Abstract |
Boerhaave’s syndrome is a rare and sometimes life-threatening condition characterized by lower esophageal tear due to a sudden rise of intraluminal pressure, and it is usually presented with a history of forceful vomiting, abdominal or chest pain, and sometimes subcutaneous emphysema. Atypical and delayed presentation is also not uncommon. A high index of suspicion is needed for diagnosis. The diagnosis is confirmed by contrast esophagogram or contrast computed tomography (CT) of the chest. Mortality rates could be as high as nearly 20-40% even in the treated cases. In this article, we report a case of a 57-year-old male, who is a smoker as well as an alcoholic, with right-sided hydropneumothorax presenting with dyspnea, epigastric pain, hoarseness of voice, and features of shock. Boerhaave’s syndrome was suspected on finding food particles in pleural fluid that contained very high salivary amylase level. The diagnosis was confirmed with a sterile methylene blue contrast study.
Keywords: Boerhaave′s syndrome, hydropneumothorax, methylene blue
| How to cite this article: Ta RK, Banerjee SN, Roy A, Pandey AK, Chatterjee K, Barma P. Boerhaave’s syndrome presented with right-sided hydropneumothorax and hoarseness of voice: A case report. Ann Trop Med Public Health 2015;8:300-3 |
| How to cite this URL: Ta RK, Banerjee SN, Roy A, Pandey AK, Chatterjee K, Barma P. Boerhaave’s syndrome presented with right-sided hydropneumothorax and hoarseness of voice: A case report. Ann Trop Med Public Health [serial online] 2015 [cited 2020 Sep 22];8:300-3. Available from: https://www.atmph.org/text.asp?2015/8/6/300/162605 |
| Introduction |
Boerhaave’s syndrome, first described by the Dutch physician Herman Boerhaave, refers to an intramural esophageal tear involving the left distal portion of the esophagus and is associated with forceful vomiting and retching, causing a rapid rise of intraluminal pressure leading to the perforation. [1] It is a rare but life-threatening condition requiring urgent diagnosis and treatment. A high index of suspicion is needed for prompt diagnosis because atypical presentation could occur in absence of the classic triad of symptoms, i.e., vomiting, abdominal or chest pain, and subcutaneous emphysema. The diagnosis is confirmed by contrast esophagogram. Delay in treatment may cause potentially lethal complications such as mediastinitis, pleural empyema, septic shock, and even multiple organ failure. The treatment of this disease is controversial based on endoscopic, surgical, or conservative mode. [2] The mortality rate is high, depending on the duration and severity of illness, and expertise of the treatment center. [2],[3]
Here, we report a case of a smoker as well as an alcoholic patient who came to us 72 h after starting abdominal pain and presented with severe breathlessness, chest pain, and hoarse voice. The case turned out to have Boerhaave’s syndrome. Clinicians should be aware of this rare, lethal disease, its unusual presentations, and the importance of its early diagnosis. We emphasize to consider this diagnosis in an acutely dyspnoeic patient with a history of gastrointestinal and respiratory complaints.
| Case Report |
A 57-year-old male patient, who was an alcoholic for the last 25 years and had a smoking history of 30 pack-years, was admitted to our ward with severe breathlessness, hoarseness of voice, and restlessness. His symptoms started initially with a sudden onset of a constant dull aching pain in the upper abdomen followed by an insidious onset of pleuritic type of pain in the right lateral chest wall. Both food intake and swallowing would increase pain. Initial self-medication with proton pump inhibitors did not help. After 3 days, he was rushed to our hospital as he developed sudden breathlessness that became severe within few hours. He also had hoarseness of voice accompanied with breathlessness. Symptoms like stridor, wheeze, or hemoptysis were absent. On his way to the hospital, he experienced a gradual diffuse swelling over the anterior chest wall with some feeling of crepitus but not over his neck. On enquiry, an episode of binge drinking followed by retching and vomiting was revealed 4 days prior to developing these symptoms. His medical history was uneventful except that of smoker’s cough. On admission, he was anxious and restless and his fingertip oxygen saturation (Sp0 2 ) was 85% in room air. A rapid clinical assessment of the patient was done with prompt institution of supplemental oxygen for severe breathlessness. General survey revealed regular respiration rate to be 36 breaths per minute and low volume regular pulse to be 130 bpm. Supine blood pressure was 70/40 mmHg. Pallor and proptosis were observed. Chest examination revealed bulging right anterior hemithorax with diminished movement. Subcutaneous crepitus was palpated over the swollen area. Both the trachea and cardiac apex were shifted to the left side. On percussion, hyperresonant note was elicited along the midclavicular line up to the right fourth intercostal space (ICS) in sitting position and also over the right supra scapular and the upper interscapular and upper axilla. A stony dullness was elicited on purcussion from right fifth ICS downward anteriorly merging with liver dullness and also the lower axillary and infrascapular areas. The topmost point of dullness along three standard lines of pucussion on the right hemithorax appeared horizontal. Shifting dullness was present but the succussion splash sound was absent. The left hemithorax was resonant with normal position of Traube’s space. Breath sound in whole of the right chest was absent. However, vesicular breath sound with prolonged expiration was detected in the left hemithorax. Heart sound was normal. Abdomen was scaphoid with epigastric tenderness and sluggish peristaltic sound. A clinical diagnosis of right-sided hydropneumothorax was made initially with suspected underlying chronic obstructive pulmonary diseases (COPD). An urgent standard chest x-ray revealed right-sided hydropneumothorax with contralateral mediastinal shift [Figure 1]. A quickly sent blood count report showed moderate anemia with a hemoglobin level 8 mg/dL and high total leukocyte count of 15,600 with 90% neutrophils; erythrocytes sedimentation rate was 100 in the first hour. His serum bilirubin was 2 mg/dl with normal liver enzymes along with low serum albumin but normal serum globulin. Serum urea level was 56 mg/dL and creatinine level was 1.3 mg/dL. Levels of serum amylase and lipase were normal that is 60 μ/dL and 56 μ/dL, respectively. Results of the straight x-ray of abdomen and electrocardiogram were normal. The thoracocentesis revealed deep color, malodorous pleural fluid with some debris. Its analysis showed leukocyte count to be 3500 cells/cmm with 95% neutrophil and 5% lymphocyte, negative for gram stain as well acid fast bacilli. Pleural fluid report also showed the presence of yeast cells, plenty of pus cells, coliform bacilli, oil droplets, few vegetable cells, and plenty of debris of food particles. Pleural fluid protein was 4 gm/dL, sugar level was 36 mg/dL, and adenosine deaminase was 200 IU/L. We suspected this case as Boerhaave’s syndrome at this stage. Further pleural fluid assay revealed very high content of amylase (263 IU/L) and salivary amylase (435 IU/L). Ultrasonography of the whole abdomen was normal. For treatment, an intercoastal chest tube (ICT) drain was put promptly to relieve severe breathlessness. A yellowish turbid fluid with some gross food particles came out by the ICT drain [Figure 2]a and b. The breathlessness of the patient was relieved to some extent but oxygen supplementation still needed. Thereafter, a contrast-enhanced computed tomogram (CECT) scan of the chest was undertaken that showed right hydropneumothorax with air in mediastinum and adjacent soft tissue subcutaneous emphysema and chest tube [Figure 3]a. A contrast study was planned at bedside with sterile methylene blue that was swallowed by the patient and it came out through the intercoastal tube quickly. Thus, the contrast test turned out to be positive [Figure 3]b, indicating the esophagopleural communication. The diagnosis was thus confirmed “Boerhaave’s syndrome”. An upper gastrointestinal endoscopy could not be done as the patient died unfortunately on the very next day.
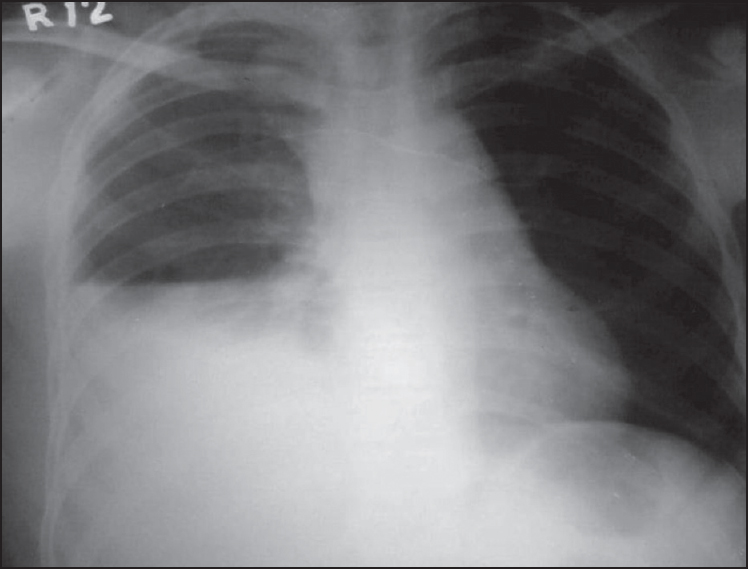 |
Figure 1: Chest x-ray (posteroanterior view) showing right hydropneumothorax and contralateral mediastinal shift
Click here to view |
| Figure 2: (a) Food particles in intercoastal tube drain, (b) pleural fluid turbid materials in water seal drain bag
Click here to view |
| Figure 3: (a) CECT scan of the thorax showing subcutaneous emphysema with hydropneumothorax with cut section of intercoastal tube, (b) the methylene blue dye coming out through ICT intercoastal chest tube
Click here to view |
| Discussion |
Boerhaave’s syndrome is a barogenic intramural tear of the esophagus caused by a sudden rise of intraluminal pressure. It accounts for approximately 15% of all cases of spontaneous esophageal rupture and has a mortality rate up to 40%. [1] The usual symptoms are vomiting, chest pain, dyspnea, mediastinal or subcutaneous emphysema, and sometimes cardiovascular collapse. [1] However, up to one-third of patients with atypical symptoms are admitted to hospitals with severe respiratory distress and/or shock, [4] as in our case. The differential diagnosis of Boerhaave’s syndrome includes a variety of acute thoracic and abdominal conditions like perforated peptic ulcer, Mallory-Weiss syndrome More Details, pancreatitis, myocardial infarction, pulmonary embolus, ruptured aortic aneurysm, pneumonia, and spontaneous pneumothorax. [4] Because the signs were nonspecific and symptoms like epigastric pain could obscure the clinical picture, delay in definitive diagnosis and treatment might occur, [1],[5] as happened in case of our patient. Our patient sufferred initially with epigastric pain and right lateral chest pain, and later developed progressive severe breathlessness, hoarseness of voice, and restlessness with features of shock. A diagnostic challenge thus existed. Presence of food particles, a high amylase content, and a low pH in pleural fluid are either highly confirmatory or highly suggestive of esophageal perforation, [3],[6] as was demonstrated in our case. Imaging study is key to confirm the diagnosis. X-ray of the esophagus with contrast is the first choice for definitive diagnosis with a false negative rate of 10-25%, while chest CT scan with oral contrast is the second choice. [1],[5],[7] The findings of mediastinal fluid and more importantly mediastinal air on CT scan of the chest are strongly suggestive of esophageal perforation. In our case, contrast study with sterile methylene blue was undertaken. And this confirmed the diagnosis of Boerhaave’s syndrome. The esophagopleural communication was shown by quick accumulation of the dye in the chest drain bag [Figure 3]a]. The dye methylene blue was cheap, easily available, and quite a suitable alternative in resource- poor rural setting(s). Our case showed several unusual features like a delayed presentation, epigastric pain, features of shock, hoarseness of voice, and underlying COPD. Breathlessness and subcutaneous emphysema appeared late. We speculated that the esophageal rupture probably did not violate the pleura or mediastinum instantaneously. Rather the overlying pleura was gradually digested by gastric contents and thus, a right-sided hydropneumothorax developed later with a marked increase in breathlessness and features of shock, which promoted the patient to come to the hospital. An epigastric pain in an alcoholic and smoker, accompanied with chest pain, might origin from peptic ulcer disease or from the many differential diagnosis as mentioned. The patient was treated initially by proton pump inhibitors without any improvement. The exact reasons of hoarseness of voice were unclear. We thought that hoarseness of voice developed because of the mediastinal shift or tractional pressure changes over adjacent mediastinal structures. The vocal cords could not be inspected for any local changes related to subcutaneous emphysematous that developed externally. Management of Boerhaave’s syndrome remained controversial. The literature suggested three strategies of treatment, i.e, endoscopic, open surgery, and conservative approach. [8],[9] De Schipper et al. proposed surgery (i.e., open thoracotomy with resection, hemi-fundoplication, and pleural/mediastinal drainage) for a patient diagnosed after 48 h with a septic profile. [2] Others recommend an aggressive treatment of sepsis and image-guided drainage of the leakage that in most cases avoids major surgery and allows esophageal healing with reduced mortality and morbidity. [10] Abbas et al. also advocated primary surgical repair when patients present with sepsis, large uncontained leaks, and extensive contamination. [11] We wanted to follow the recommendations of de Schipper et al. in our case because of late diagnosis and septic features. Unfortunately, esophagoscopy and thoracic surgery could not be done as the patient died on that very day. Despite treatment, depending on the duration, severity, and expertise of the treatment center, mortality rates are nearly as high as 20-40% in treated and 100% in untreated cases. [2],[3] Survival is directly related to the time of diagnosis and treatment, a high clinical suspicion can decrease the substantial rates of morbidity and mortality associated with Boerhaave’s syndrome. [12]
| Conclusion |
Boerhaave’s syndrome is an uncommon clinical entity with variable modes of presentation, making the diagnosis challenging. Diagnosis and treatment should be prompt. The diagnosis should be considered in patients with a combination of gastrointestinal and respiratory complaints. A methylene blue dye test could be used for a contrast imaging study to confirm the diagnosis, especially in rural setting(s). Outcomes could turn out to be fatal with late presentation and features of shock.
| Acknowledgment |
General support from all the concerned staff of our department are gratefully acknowledged.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| References |
| 1. |
Janjua KJ. Boerhaave’s syndrome. Postgrad Med J 1997;73:265-70.
|
| 2. |
de Schipper JP, Pull ter Gunne AF, Oostvogel HJ, van Laarhoven CJ. Spontaneous rupture of the oesophagus: Boerhaave’s syndrome in 2008. Literature review and treatment algorithm. Dig Surg 2009;26:1-6.
|
| 3. |
Curci JJ, Horman MJ. Boerhaave’s syndrome: The importance of early diagnosis and treatment. Ann Surg 1976;183:401-8.
|
| 4. |
Brauer RB, Liebermann-Meffert D, Stein HJ, Bartels H, Siewert JR. Boerhaave’s syndrome: Analysis of the literature and report of 18 new cases. Dis Esophagus 1997;10:64-8.
|
| 5. |
Lemke T, Jagminas L. Spontaneous esophageal rupture: A frequently missed diagnosis. Am Surg 1999;65:449-52.
|
| 6. |
Henderson JA, Péloquin AJ. Boerhaave revisited: Spontaneous esophageal perforation as a diagnostic masquerader. Am J Med 1989;86:559-67.
|
| 7. |
Backer CL, LoCicero J 3 rd , Hartz RS, Donaldson JS, Shields T. Computed tomography in patients with esophageal perforation. Chest 1990;98:1078-80.
|
| 8. |
Wolfson D, Barkin JS. Treatment of Boerhaave’s syndrome. Curr Treat Options Gastroenterol 2007;10:71-7.
|
| 9. |
Carrott PW Jr, Low DE. Advances in the management of esophageal perforation. Thorac Surg Clin 2011;21:541-55.
|
| 10. |
Vogel SB, Rout WR, Martin TD, Abbitt PL. Esophageal perforation in adults: Aggressive, conservative treatment lowers morbidity and mortality. Ann Surg 2005;241:1016-23.
|
| 11. |
Abbas G, Schuchert MJ, Pettiford BL, Pennathur A, Landreneau J, Landreneau J, et al. Contemporaneous management of esophageal perforation. Surgery 2009;146:749-56.
|
| 12. |
Jagminas L, Silverman RA. Boerhaave’s syndrome presenting with abdominal pain and right hydropneumothorax. Am J Emerg Med 1996;14:53-6.
|
Source of Support: None, Conflict of Interest: None
| Check |
DOI: 10.4103/1755-6783.162605
| Figures |
[Figure 1], [Figure 2], [Figure 3]



